Graciously
borrowed from: Clavius.org
ENVIRONMENT
radiation primer
Radiation is a hard-working word in physics. It describes
several diverse natural processes and their effects. As used in common speech,
it means what physicists call "ionizing radiation", or that which can
produce detrimental effects in materials and organic tissue. Ionization is the
process of removing electrons from atoms, and when this occurs in biological
tissues it disrupts the delicate chemical and physical processes that sustain life.
This can happen through mutation, when the DNA of the organism is altered, or
directly via the destruction of atomic bonds and the breakup of important
molecules at the site of the ionization.
A BASIC DIVISION
We consider two broad categories of ionizing radiation: that
caused by electromagnetic rays, and that caused by high-energy charged
particles.
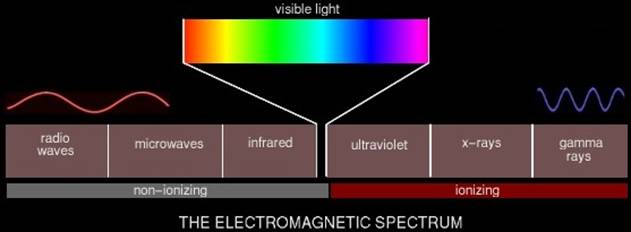
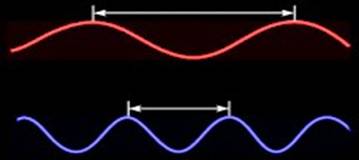
The electromagnetic spectrum is familiar to most people.
What we call "light" is really a narrow band in a single phenomenon
which includes radio waves, microwaves, and x-rays. A wave's position in the
spectrum depends on its wavelength, the distance between two adjacent
"crests" of the wave (Fig. 1). On the left are "long" waves
such as radio, television, microwave, and infrared. On the right are "short"
or "high frequency" waves such as x-rays and gamma rays.
|
|
|
|
As you can see, not all electromagnetic (EM) waves are
ionizing radiation. Generally anything above the visible spectrum is considered
ionizing radiation and thus harmful to some degree. Ultraviolet radiation from
the sun is what sometimes causes skin cancer. X-rays and gamma rays are
produced by nuclear reactions -- atomic bombs, and to a much lesser degree,
nuclear reactors.
Non-ionizing EM radiation can still be dangerous, of course,
in sufficient quantities. Microwaves cook food by exciting the water molecules
in the food until they vibrate and create heat. Obviously they can also
excite the water molecules in the human body and cause a similar effect.
The other category of ionizing radiation comprises
high-energy charged particles.

An alpha particle is the nucleus of a helium atom, composed
of two protons and two
neutrons. It has a charge of +2 and is very large and heavy.
A beta particle is an electron emitted from the nucleus of a
radioactive substance. It has a charge of -1 and is much, much less massive
than a proton or a neutron.
A proton is, well, a proton. And neutrons are neutrons.
Protons and neutrons have about the same mass, but the neutron doesn't have a
charge while the proton has a charge of +1.
Now having made a careful distinction between waves and
particles, we note that many authors use the terms interchangeably (e.g., beta
ray and beta particle). Since EM radiation is carried by the photon (a
particle) and since equivalent energies can be computed for proper particles,
there isn't any real need to maintain such a strict distinction. In fact, it's
frequently useful to be able to share measurements between all the different
kinds of radiation.
But when computing radiation dosage (the effect of radiation
on organisms) and when constructing shielding, the differences must be clearly
understood. Pound for pound, particle radiation is much more dangerous than
wave radiation. The bigger the particle, the more damage it is capable of
doing.
ENERGY AND FLUX
We mentioned above that physicists deal with radiation in a
more abstract concept. In their terminology, it is one of the mechanisms by
which energy is transferred from one place to another. Radiation is
"energy in transit". When we speak of the "energy" of a
wave we consider its intensity. The energy of a particle can be thought of as
equivalent to its speed. High-energy particles travel very fast, while
low-energy particles travel slowly.
Physicists use another measurement, "flux", to
describe a sort of particle density. If many particles pass by a certain point
in a given length of time, we say the flux is high. If few particles pass, the
flux is low. "Flux" is the Latin word for "flow".
If we take a cubic meter of space anywhere in the universe,
we'll discover that it contains many particles of varying flux and energy. In
general, flux and energy vary inversely. That means the higher the energy, the
lower the flux. So if we look at the low-energy particles, we may find an
enormous flux.
WHERE RADIATION COMES FROM
We can answer this question in two ways. We can say that
charged particles come from the nuclei of various atoms that undergo nuclear
decay. We can say that EM rays (especially x-rays and gamma rays) are emitted
from those same nuclei, and we can note that any substance with sufficient
energy, or heat,
emits EM radiation as a method of releasing that energy. That describes the
source of radiation at the microscopic level.
But the pressing issue is where in the universe we might
expect to encounter these types of radiation, and in what quantities. The short
answer is that radiation is all around us. EM radiation bombards us constantly,
but thankfully not generally in the ionizing range of wavelengths. High-energy
charged particles rain down on us from space, and are produced by the natural
radioactive decay of many natural substances. The constant low level radiation
which we encounter every day is "background radiation".
Predictably, the chief source of all kinds of radiation in
space is the sun. A full spectrum of EM waves radiates outward from it. Charged
particles of all types emanate from it, especially during periods of extreme solar activity
(e.g., flares).
Earth's atmosphere protects us from most ionizing
electromagnetic radiation from the sun. Ultraviolet, x-ray, and gamma rays
penetrate to some extent (enough to give us sunburns, for example), but in
space there is a consistently higher level of all of these. But only during
periods of extreme solar activity does this radiation exceed our ability to
shield against it.
Alpha and beta particles and protons carry electromagnetic
charges, making them susceptible to magnetic fields. The earth's magnetic field
deflects the flow of these particles from the sun. But it also causes them to
collect in two large regions of space surrounding the earth -- the Van Allen
belts. We are reasonably safe inside the Van Allen belts. And as long as the
sun remains reasonably quiet, we are even safe outside them.
|
|
|
|
But when the sun acts up, the area outside the Van Allen
belts becomes thick (i.e., high flux) with dangerous, high-energy charged
particles. A solar event was depicted in the motion picture Red Planet,
forcing the crew of that mission to seek cover.
But since the Van Allen belts themselves contain
concentrations of charged particles, going through them presents its own hazard.
We can think of it as crossing a barbed-wire fence: the fence offers
protection, but can also snag us as we crawl through it.
We've left neutrons out of the picture up until now, and
that's because they just don't occur as high-energy particles anywhere
in the universe in numbers great enough to care about. Scientists even have
trouble creating them in the lab.
HOW TO SHIELD AGAINST RADIATION
This is where the difference between radiation types becomes
important. Wave radiation requires thick, heavy shielding. It requires
considerably less material to block particles.
In general, the shorter the EM wavelength, the thicker and
denser the shield material must be. Ultraviolet (UV) can be blocked simply by a
sufficiently opaque sheet of plastic. We are all familiar with tinted
sunglasses that promise to block some 97% of solar UV rays. Not much additional
protection is required in space. X-rays and gamma rays are another matter.
Where intense x-rays and gamma rays occur, it requires several inches or
centimeters of lead and/or concrete to provide adequate shielding.
Alpha particles are very large particles. As such they don't
penetrate very deeply into many things. In fact, alpha particles will not even
penetrate the epidermal (dead) layer of skin, and so present no special hazard
to humans. A sheet of reasonably thick paper will block all alpha particles.
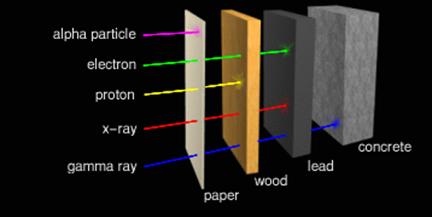
Protons penetrate farther. They can be shielded by light
metals or plastics in thicknesses of about a centimeter.
Beta particles are very small and can penetrate centimeters
into the body. But luckily they're too small to cause much damage if they hit
anything. But there's a special problem here. When beta particles hit large
atoms, the impact causes those atoms to give off x-rays. Metal atoms are
usually quite heavy, and so are especially susceptible to this kind of
re-radiation which is known by its German name "Bremsstrahlung". In
fact, this is how x-rays are produced intentionally for medical applications.
The best materials to shield against beta particles have
lots of hydrogen atoms in them. Hydrogen atoms are light, and so absorb the
particles without giving off x-rays. Plain old water works very well. In fact,
4 inches (10 centimeters) of water will block almost all background beta
particles. But water is impractical for shielding in space, so high-density
polyethylene (HPDE, chemical formula CH2CH2...) is
frequently used instead. This also effectively blocks protons.
AN ALTERNATIVE TO SHIELDING
Radiation exposure is cumulative, meaning that the longer
you're exposed to it, the worse effect it has. It's very much like running
through the rain. We've discussed shielding, which is like an umbrella. But if
it's impractical to provide complete shielding, you can also reduce the
exposure time. This is the same as running through the rain rather than
walking.
If you forget your umbrella on a rainy day and have to park
some distance from your destination, you can reduce your "exposure"
to the rain by running from the car to the door. If you walk instead, you'll
spend more time under the rain and thus get wetter.
Organisms can recover from exposure to radiation, just as
you can eventually dry after walking or running through the rain. The wetter
you get, the longer it takes to dry. The more exposure to radiation, the harder
it is to recover. The body will repair damage done to DNA or to other important
molecules, although it will be sick in the meantime. It's actually better to
absorb a high dose of radiation quickly than a low dose over a long period.
Although the higher dose may cause more problems in the short term, the low
dose will produce continuing damage and your body simply may not be able to
keep up even though the damage is slight at any one moment.
When it comes to designing space ships, additional shielding
means additional weight, and that means your space ship may have to go slower.
The answer to this tradeoff is to skimp on shielding and go faster. The
radiation exposure will be more intense, but it will not last as long. This is
preferred.
HOW TO MEASURE RADIATION
Most people aren't familiar with the various units and
concepts used to measure radiation. It just isn't something they have to deal
with. And so when conspiracists describe radiation using big numbers they find
in textbooks and elsewhere, the general public isn't always equipped to
understand what those numbers mean.
The problem is exacerbated by the fact
that Americans have one system of units for measuring things, and the rest of
the world has another system. This is also true for radiation. So not only do
people have to deal with labels they've never seen before, but they don't know
what measurements are simply differences in units, a sort of radiological
furlong versus a radiological centimeter
We measure two general phenomena when we discuss radiation.
We measure "activity" and "exposure". Activity is basically
just how much radiation is coming out of something, whether it's particles or
waves. Exposure is the important factor. It measures the effect of radiation on
substances that absorb it.
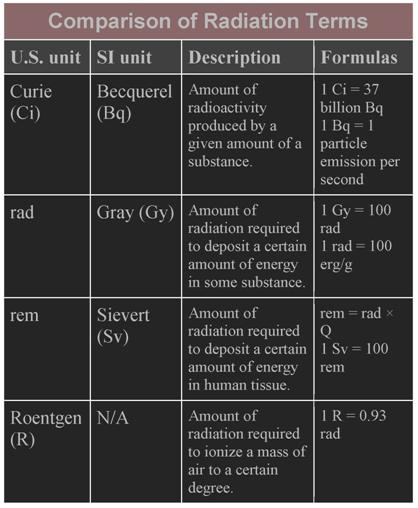
Radiation activity is measured in an American unit called a
"Curie" (Ci) or an international (SI) unit called a
"Becquerel". The Curie is defined by how much radiation one gram of a
radium isotope emits. The Becquerel just counts how many particles or photons
(in the case of wave radiation) are emitted per second. The device used for measurement
is often the familiar Geiger counter. If you put a Geiger counter over a gram
of substance and count 3 clicks per second, the radioactivity of that substance
would be 3 Bq.
Radiation exposure is measured in
American units by the "rad", an acronym standing for "radiation
absorbed dose", and in the SI system by the Gray (Gy). The
exposure is the amount of energy "deposited" in a substance by
radiation. A rad is the amount of radiation required to deposit 100 ergs of
energy in a gram of material. An erg is a very small amount of energy, but it
takes only a very small amount of energy to ionize an atom. The number isn't
important. The important concept is that exposure is measured by
what radiation does to substances, not anything particular
about the radiation itself. This allows us to unify the measurement of
different types of radiation (i.e., particles and wave) by measuring what they
do to materials.
But what materials? Wood, water, human tissue -- they all
have different densities, so a gram of one material may be bigger or smaller
than a gram of another material. And the bigger something is, the more surface
area is available for bombardment by rays or particles. It would be nice if
there were some way of comparing exposure in various substances directly.
Enter the "rem". That's another acronym, meaning
"radiation equivalent, man". As with all
measurements of exposure it describes the effects of radiation on substances
that absorb it, but in this case the substance is specifically human tissue.
It's an American unit; the corresponding SI unit is the Sievert (Sv).
The reader is likely to encounter the term
"Roentgen", which is another American unit of exposure. It measures the
amount of ionization a certain amount of radiation produces in air, and has
been largely abandoned in favor of the rad. It can be roughly equated to a rad
for estimation purposes.
Above we discussed that different kinds of radiation are
inherently more dangerous than others. By measuring exposure in how it affects
surfaces, we can largely ignore the differences in kinds of radiation. But in
order to compute rems from rads we need to take into account that some kinds of
radiation are inherently more dangerous to biological tissue, even if their
"energy deposition" levels are the same. Each kind of radiation
carries a "relative biological effectiveness" (RBE) factor, also
called a "quality factor" (Q).
For x-rays and gamma rays and electrons absorbed by human
tissue, Q is 1. For alpha particles it is 20. For protons and neutrons, it is
10. To compute rems from rads, or Sieverts from Grays, simply multiply by Q.
This is obviously a simplification. The RBE/Q factor approximates what
otherwise would be very complicated computations. And so the values for Q
change periodically as new research refines the approximations.
Exposure occurs over time, of course. The more rems absorbed
in a unit of time, the more intense the exposure. And so we express actual
exposure as an amount over a specific time period, such as 100 rads per hour,
or 5 millisieverts per year. This is called the "dosage rate", and is
proportional to the flux of radiation in a particular situation.
HOW MUCH IS TOO MUCH?
Conspiracy theorists exploit the natural radiophobia that
has arisen since the bombing of Japan with nuclear weapons, testing by various
nations, and the Chernobyl accident in the former Soviet Union. But now that we
understand a little bit about how radiation is measured, we can quantify the
danger.
The U.S. government endorses the recommendations of various
international regulatory bodies on the acceptable levels of radiation exposure
in the workplace and among the general public.
If a worker must deal with radioactive materials in the
course of his job, his legal limit is higher: 5 rem (50 millisieverts, mSv) per
year. If a worker is in the vicinity of radioactive materials but does not work
with them, the limit is 0.1 rem (1 mSv). For persons younger than 18 and
pregnant women, the occupational exposure is 0.5 (5 mSv) per year. These are
measurements above the natural background radiation limits, and are measured by
dosimeters and other equipment in the area where the exposure takes place. (Standards
for Protection Against Radiation. 10 CFR § 20.)
People usually get about 0.24 rem (2.4 mSv) in background
radiation per year. (Jawororwski, Zbigniew. "Radiation Risks in the 20th
Century: Reality, Illusions, and Risks" Presented 17 Sept. 1998 at the
International Curie Conference, Warsaw, Poland.)
The standard for a lethal dose is designated LD 50/30,
defined as the short-term exposure (i.e., over a period of a few hours or less)
which would kill 50% of the human population within 30 days. It's around
350-400 rems (3.5-4.0 Sv). (Radiation Safety Office. Radiation Safety
Handbook. Columbia University, s.d.)
The limits imposed by U.S. Federal
Regulations are thus extremely conservative. The lethal dose is 700 times the
amount of radiation acceptable per year for people who work around
radioactivity. The regulations are so very strict because while it has been
determined that even dosages up to 30 rems per year produce no visible effect,
there is no such thing as radiation with no harmful effects. It just happens
that for low doses the body can repair itself effectively.